The AccessDx PGx Profile:
PGx for Oncology
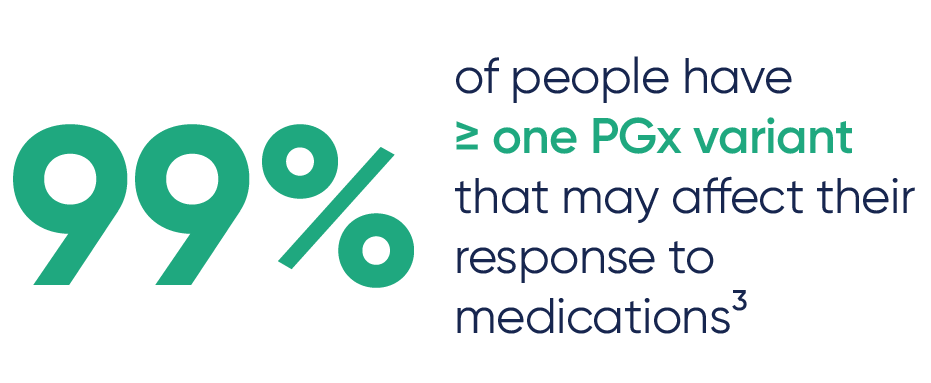
PGx testing helps oncology teams personalize cancer treatment by identifying genetic differences that can affect how a patient metabolizes and responds to key oncology and supportive medications.
- Helps reduce serious adverse drug reactions
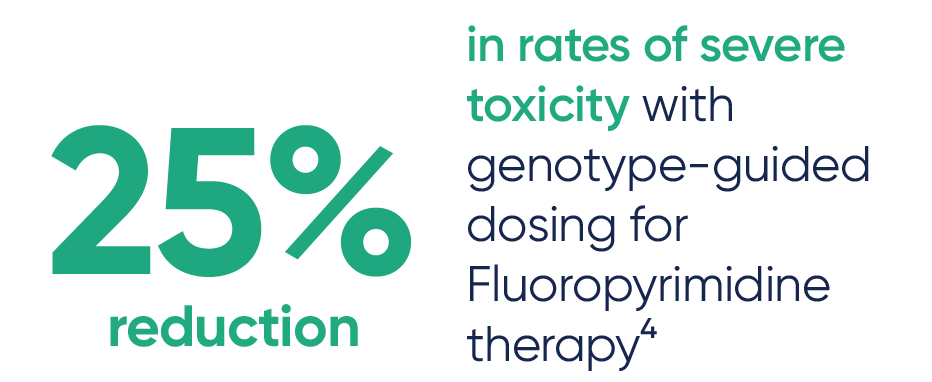
- Reduces the risk of severe toxicity
- Improves treatment tolerance
- Enables more personalized cancer care
- Provides actionable insights before treatment begins
Fill out this form to learn more details about PGx use in Oncology practices.
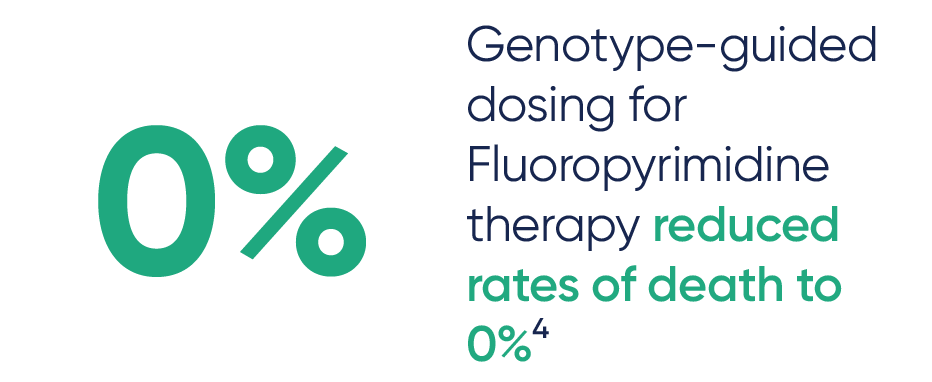
PGx testing can guide dosing for some chemotherapy medications like Capecitabine, Fluorouracil, and Irinotecan, and can help identify patients who may not benefit from Tamoxifen2
Testing Offerings:
Therapeutic Areas Impacted:
Oncology
Behavioral Health
Gastroenterology
Hyperuricemia Management
Infection Management
Neurology
Pain Management
Rheumatology & Autoimmune Disorders
Urology
Resources:
Government and Scientific Resources
AccessDx PGxperts™ Articles
- Journal of Clinical Pathways: Clinical Implications of the FDA’s Capecitabine Boxed Warning: Integrating DPYD Testing into Oncology Practice
- Healthcare Business Today: Pharmacogenomics in Oncology: Unlocking Value, Safety, and ROI for Healthcare Systems
- MedCity News, Drug-Gene Testing: Key to Safer Cancer Treatment
- Journal of Clinical Pathways, FDA Highlights Risks Associated With DPD Deficiency, Advises Testing Before Treatment
- Pharmaceutical Executive, Screening for DPD: Q&A with Kristine Ashcraft
- Journal of Clinical Pathways, “Implementing PGx Testing in Oncology: Best Practices, Barriers, and Benefits.”
- Journal of Clinical Pathways, “Recommendations for Standardizing DPYD Pharmacogenomic Testing.”
- Ronquillo JG, Lester WT Pharmacogenomic testing and prescribing patterns for patients with cancer in a large national precision medicine cohort Journal of Medical Genetics 2023;60:81-83.
- U.S. Food and Drug Administration. “Table of Pharmacogenetic Associations.” FDA, 26 Oct. 2022, www.fda.gov/medical-devices/precision-medicine/table-pharmacogenetic-associations. Accessed 8 Apr. 2025.
- Chanfreau-Coffinier C, et al. Projected Prevalence of Actionable Pharmacogenetic Variants and Level A Drugs Prescribed Among US Veterans Health Administration Pharmacy Users. JAMA Netw Open. 2019;2(6):e195345.
- Deenen, Maarten J et al. “Upfront Genotyping of DPYD*2A to Individualize Fluoropyrimidine Therapy: A Safety and Cost Analysis.” Journal of clinical oncology : official journal of the American Society of Clinical Oncology vol. 34,3 (2016): 227-34. doi:10.1200/JCO.2015.63.1325


